THUNDER BAY – Mining – Zenyatta Ventures Ltd. (TSX VENTURE:ZEN) is reporting on recent geological research at Lakehead University in Thunder Bay. The report covers Zenyatta’s unique Albany graphite deposit.
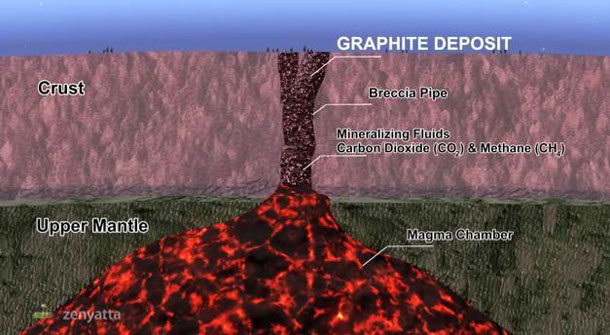
The unusual mode of formation, through igneous hydrothermal processes, accounts for the superior purity and crystallinity of graphite found in this ‘one of a kind’ deposit.
The Government of Canada has awarded a collaborative research grant through the Natural Sciences and Engineering Research Council (‘NSERC’) to assist Dr. Andrew Conly (associate professor, Department of Geology at Lakehead University) in carrying out further studies on the unique Albany graphite deposit. This is in addition to the support Zenyatta recently received from the National Research Council of Canada Industrial Research Assistance Program for metallurgical testing.
Dr. Andrew Conly observed, “Evidence has shown that Zenyatta has discovered a unique sub-class of a hydrothermal graphite deposit unlike any other. Igneous breccia-hosted graphite deposits like Albany are very rare, and to the best of my knowledge, none are currently being mined or even in an advanced stage of exploration globally. Our on-going research of the Albany deposit will establish the first genetic model for this distinctive type of graphite. I would like to thank NSERC for their recent important contribution in assisting myself and students at Lakehead University during further development of this geological model.”
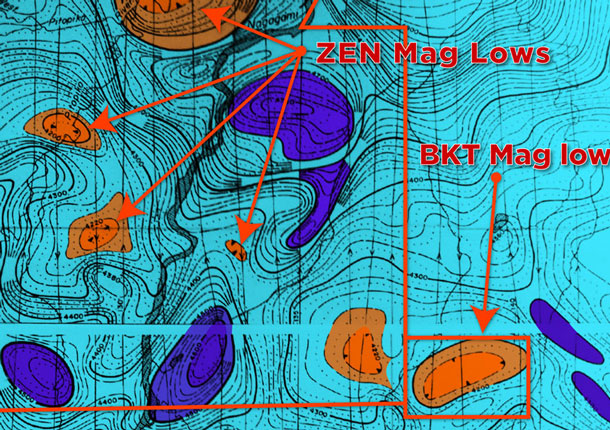
Diagrams showing the various stages of geological deposition can be found at www.zenyatta.ca under ‘Graphite Project’.
Crystalline Graphite – High Tech Applications
In contrast to more commonly occurring flake and amorphous graphite deposits, the unusual hydrothermal style in the Albany deposit can be processed, at a cost advantage, to yield high purity, crystalline graphite ideally suited for advanced high-tech applications. The world trend is to develop products for technological applications that need extraordinary performance using ultra-high purity graphite powder at an affordable cost. High purity is gaining prominence at a time when Zenyatta discovered a very rare, (hydrothermal) graphite deposit, which can be upgraded to 99.99% carbon (‘C’) with very good crystallinity without the use of aggressive acids and high thermal treatment. The development of this deposit would place Zenyatta in a strong position to compete in specialised markets such as those currently supplied by high-cost synthetic graphite. When combined with a large, discrete ultra-high purity graphite deposit and the growth potential of these markets, the substantial potential of the Albany graphite deposit becomes quite evident.
Dr. Conly stated, “The preliminary genetic model (as presented in the NI 43-101 Technical Report, Jan 2014) had to account for Albany’s key attributes, including: 1) highly pure and crystalline graphite; 2) brecciated (volcanic-hosted) pipes, consisting of both angular & rounded fragments; and, 3) simple mineralogy (i.e. essentially graphite, quartz & feldspar) with minimal alteration associated with graphite deposition. Our research at Lakehead University is working on characterizing the nature of the graphite forming fluid (i.e. carbon dioxide CO2 versus methane CH4). Such information is crucial for developing a comprehensive and well-constrained genetic model for this uncommon type of graphite mineralization.”
Dr. Conly also noted that, “The far more common flake type (sedimentary) graphite deposits form through a completely different geological process. Flake graphite commonly occurs globally in metasedimentary rocks as a result of the conversion of organic matter through regional or contact metamorphism. Graphitization of organic matter is well understood to be due to heating and compression of organic matter in situ over a long period of time and this process results in graphite that contains various impurities.”
Peter Wood, VP Exploration for Zenyatta, commented, “Dr. Conly has been involved in the study of the Albany graphite deposit since Zenyatta’s initial discovery drill hole in 2011. He was one of the first to recognize the geological significance and the economic importance of such a find. Academic work completed under Dr. Conly’s supervision has considerably increased our understanding of the geological processes required for formation, and confirms that the Albany graphite deposit is not like any other graphite occurrence previously discovered or documented. It represents a unique geological setting that has led to the formation of highly crystalline graphite of unusually high purity.”
A high degree of crystallinity and purity results in various positive qualities that graphite is known for such as electrical conductivity, thermal conductivity, compressibility, dimension stability, bending strength and lubricity. These qualities are very important for emerging high-tech or clean-tech applications. The highest quality material of ultra-high purity (greater than 99.95% C) graphite will demand a premium price in the range of $5,000 – $25,000 per tonne depending on the application.
Others like synthetic graphite producers and upgraded flake producers (including China) are faced with escalating energy costs associated with turning petroleum (needle) coke or flake graphite into high purity powder. It requires extensive thermal treatment (up to 3000 degrees C) and/or aggressive acid treatment. Not only is this a costly process for others but it also has associated environmental issues.

Types of Graphite
Somewhat simplified, there are three (3) different processes leading to the formation of graphite deposits. Natural graphite material has varying levels of quality depending on the type (amorphous, flake or hydrothermal). The degree of purity can vary greatly, which heavily influences the use of the material in applications and its pricing:
1. Amorphous (Sedimentary) graphite is derived from the metamorphism of coal deposits. Graphite formed under these conditions is characterized by incomplete structural ordering, abundant impurities and low crystallization, resulting in low value “amorphous” graphite with its main market in foundry applications.
2. Flake (Sedimentary) graphite is the metamorphism of organic material. The formation of these deposits involves sedimentation and then alteration of carbonaceous organic matter to graphite during regional metamorphism containing abundant impurities. Upgrading of graphite from this deposit type is complex and costly as a result of processing using aggressive acids and/or thermal treatment.
3. Hydrothermal (Volcanic) graphite deposits. The formation of these deposits is associated with migrating supercritical carbon-bearing (C-O-H) fluids or fluid-rich magmas associated with igneous (or volcanic) activity. The formation of the carbon-bearing fluids is most often a consequence of high temperature metamorphism, but magmatic degassing can also produce graphite. Fluid precipitated graphite is well-ordered and can be a source of highly valued crystalline or vein-type graphite.
The Albany graphite deposit is a unique example of a hydrothermal graphite deposit in which a large volume of highly crystalline, fluid-deposited graphite occurs within an igneous host. The deposit is interpreted as a vent pipe breccia that formed from CO2-rich fluids that evolved due to pressure-related degassing of syenites of the Albany Alkalic Complex (Conly, 2014). A more detailed explanation can be found in the NI 43-101 Technical Report dated 16 January 2014 under the section ‘Deposit Type’ filed on www.sedar.com.
Zenyatta continues to develop its 100% owned Albany Graphite Deposit located in northeastern Ontario, Canada. The Company’s graphite deposit is located 30 km north of the Trans-Canada Highway, power line and natural gas pipeline near the communities of Constance Lake First Nation and Hearst. A rail line is located 70 km away with an all-weather road approximately 4-5 km from the graphite deposit.
The outlook for the global graphite market is very favourable with demand growing rapidly from new applications. It is now considered one of the more strategic elements by many leading industrial nations, particularly for its growing importance in high technology manufacturing and in the emerging “green” industries such as electric vehicle components and energy storage. The application for graphitic material is constantly evolving due to its unique chemical, electrical and thermal properties. It maintains its stability and strength under temperatures in excess of 3,300 degrees C and is very resistant to chemical corrosion. It is also one of the lightest of all reinforcing elements and has high natural lubricating abilities. Some of these key physical and chemical properties make it critical to modern industry.